Unstable gene sites are concentrated in Autism associated genes
Why this study matters!
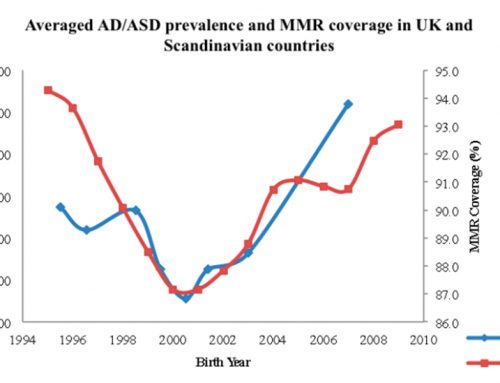
Study of autism spectrum disorder (ASD) on an individual basis has identified hundreds of diverse de novo mutations, deletions, and duplications present in about 10% of cases of simplex ASD, with an estimate of 15% when taking into account the inability to measure smaller copy number variations (CNVs).
Multiplex ASD and unaffected patients carry much smaller percentages, indicating environmental influences on the genetics of ASD. These factors point at mutations as a primary cause of autism (Sabat et. al. 2007).
Although ASD is highly heterogeneous in terms of genes5 and mutations1, the affected genes associate within biological networks, explaining the common phenotype from hundreds of diverse mutations (Gilman et. al. 2011).
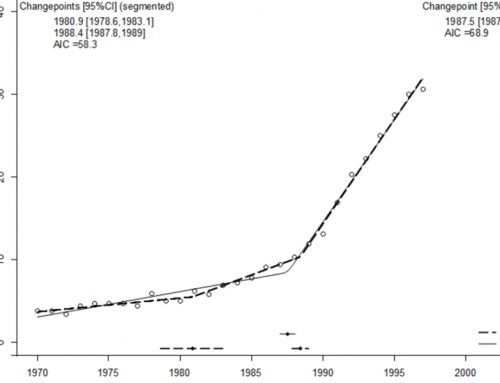
A more urgent question to be posed is “What causes hundreds of diverse de novo mutations to arise?” Altered double-strand break (DSB) formation and repair pathways may be a commonality among the diverse genetic mutations that have been documented in ASD. Much of what is known about DSB comes from the study of meiotic recombination (MR). A degenerate 13 mer sequence, CCNCCNTNNCCNC, has been associated with 41% of MR hotspots as well as with disease-causing nonallelic homologous recombination hotspots and common mitochondrial deletion hotspots (Myers et. al. 2008). The zinc finger protein, PRDM9, has been shown to be a global regulator of MR hotspots through its binding to the 13 mer consensus sequence (Berg et. al. 2010). Despite this, PRDM9 allelic expression patterns do not predict actual MR rates on a population basis (Kong et. al. 2010).
However, the relationship between MR activity and PRDM9 allelic binding affinity to constrained 13 mer sequences has never been examined, and this relationship may reveal population based associations. Sites of HR are susceptible to subsequent DSB and mutations (Hicks et.al 2010), and therefore are a potential player in the hundreds of diverse de novo mutations documented for simplex ASD.
Therefore, we have determined the genomic locations of constrained 13 mers, modeled PRDM9 binding affinity to the constrained 13 mers, and looked for associations between these DSB regulating factors and genes associated with ASD.